 | |
| Clinical data | |
|---|---|
| Routes of administration | oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
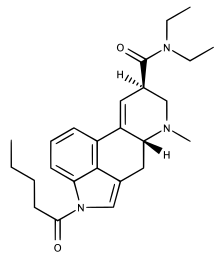
| Formula | C25H33N3O2 |
| Molar mass | 407.558 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
1V-LSD (1-valeryl-D-lysergic acid diethylamide), sometimes nicknamed Valerie, is a psychotropic substance and a research chemical with psychedelic effects.[1][2] 1V-LSD is an artificial derivative of natural lysergic acid, which occurs in ergot alkaloids, as well as being an analogue of LSD.[3] 1V-LSD has been sold online until an amendment to the German NpSG was enforced in 2022 which controls 1P-LSD and now 1cP-LSD, 1V-LSD and several other lysergamides.[4]
Pharmacology
As demonstrated with other N-acylated derivatives of LSD, 1V-LSD is believed to serve as a prodrug for LSD but may also act as a weak partial agonist at the 5-HT2A receptor.[3]
Animal studies
A Head-twitch response assay in mice found that 1V-LSD has a similar potency to 1P-LSD and 1cP-LSD, with behavioral effects also closely resembling these structural analogs.[5]
Chemistry
1V-LSD is the condensation product of valeric acid (pentanoic acid) and LSD, where the valeroyl group is substituted on the NH position of the indole moiety.[6] Ehrlich's reagent is used to identify the presence of an indole moiety; the chemical backbone of the lysergamide and ergoline molecules.[7] However, as with other N-acylated lysergamides, 1V-LSD reacts very slowly to Ehrlich reagent and may not give reliable results if the reagent isn't fresh.[8][9]
Legal position
1V-LSD is illegal in the United States as a Schedule 1 controlled drug or controlled substance under the Federal Analogue Act except if is sold for research and medical purposes like a research chemical. [citation needed]
Since March 2nd 2022, 1V-LSD has been under investigation in Sweden and may therefore soon become controlled.
1V-LSD was placed under legal control in South Korea in July 2022 on a temporary but renewable basis.[10]
An amendment to the NpSG banned the sale of 1V-LSD in Germany in September 2022. Due to a interpunctation error in the actualised NpSG, the ban never took effect.[11] The law was amended in March 2023, now banning 1V-LSD.

See also
References
- ^ "1V-LSD (solution)". www.caymanchem.com.
- ^ "1V-LSD PsychonautWiki". Psychonautwiki.
- ^ a b Brandt SD, Kavanagh PV, Westphal F, Pulver B, Morton K, Stratford A, et al. (November 2021). "Return of the lysergamides. Part VII: Analytical and behavioural characterization of 1-valeroyl-d-lysergic acid diethylamide (1V-LSD)". Drug Testing and Analysis. 14 (4): 733–740. doi:10.1002/dta.3205. PMC 9191648. PMID 34837347.
- ^ "1V-LSD - legales LSD 3.0". YouTube (in German).
- ^ Halberstadt AL, Chatha M, Klein AK, McCorvy JD, Meyer MR, Wagmann L, Stratford A, Brandt SD (August 2020). "Pharmacological and biotransformation studies of 1-acyl-substituted derivatives of d-lysergic acid diethylamide (LSD)". Neuropharmacology. 172: 107856. doi:10.1016/j.neuropharm.2019.107856. PMC 9191647. PMID 31756337.
- ^ Umehara A, Ueda H, Tokuyama H (November 2016). "Condensation of Carboxylic Acids with Non-Nucleophilic N-Heterocycles and Anilides Using Boc2O". The Journal of Organic Chemistry. 81 (22): 11444–11453. doi:10.1021/acs.joc.6b02097. PMID 27767302.
- ^ De Faubert Maunder MJ (August 1974). "A field test for hallucinogens: further improvements". The Journal of Pharmacy and Pharmacology. 26 (8): 637–8. doi:10.1111/j.2042-7158.1974.tb10677.x. PMID 4155730. S2CID 97915487.
- ^ "REACTIONS 1V-LSD". 16 July 2021.
- ^ "1V-LSD reaction with the ehrlich reagent". 16 July 2021.
- ^ Kim Chan-hyuk, Regulator names 1V-LSD as narcotic drug temporarily. Korea Biomedical Review, 5 July 2022
- ^ "Gesetzespanne: Gefährliche LSD-Derivate plötzlich legal".
| Lysergic acid derivatives |
|
|---|---|
| Psychedelic lysergamides |
|
| Clavines | |
| Other ergolines | |
| Natural sources |
Morning glory: Argyreia nervosa (Hawaiian Baby Woodrose), Ipomoea spp.(Morning Glory, Tlitliltzin, Badoh Negro), Rivea corymbosa (Coaxihuitl, Ololiúqui) |