-
1gdc: Refinirano rešenje strukture DNK vezujućeg domena glikokortikoidnog receptora
-
1glu: Kristalografska analiza interakcija glikokortikoidnog receptora sa DNK
-
1m2z: Kristalna struktura dimernog kompleksa ljudskog ligan vezujućeg domena glukokortikoidnog receptor vezanog za deksametazon i TIF2 koaktivatorski motiv
-
1nhz: Kristalna struktura antagonistne forme glukokortikoidnog receptora
-
1p93: Kristalna struktura agonistne forme glikokortikoidnog receptora
-
1r4o: Kristalografska analiza interakcije glukokortikoidnog receptora sa DNK
-
1r4r: Kristalografska analiza interakcije glukokortikoidnog receptora sa DNK
-
1rgd: Refinirano rešenje strukture DNK vezujućeg domena glikokortikoidnog receptora iz NMR podataka putem proračuna relaksacione matrice
-
2gda: Refinirano rešenje strukture DNK vezujućeg domena glikokortikoidnog receptora
| Nuklearni receptor potfamilije 3, grupa C, član 1 (glukokortikoidni receptor) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
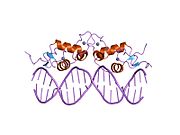
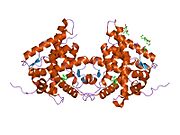
 Kristalografska struktura DNK vezujućeg domena glukokortikoidnog receptora (DBD, levo, 1R4O vezan za DNK) i ligand vezujući domen [LBD, desno, 1M2Z vezan za deksametazon (beli štapići) i TIF2 koaktivatorski protein (crveno)]. Isprekidane žute linije predstavljaju interakcije vodoničnog vezivanja između receptora i liganda. 2D struktura deksametazona je takođe prikazana u dole desno. | |||||||||||
| Dostupne strukture | |||||||||||
| 1M2Z, 1NHZ, 1P93, 3BQD, 3CLD, 3E7C, 3H52, 3K22, 3K23, 4HN5, 4HN6 | |||||||||||
| Identifikatori | |||||||||||
| Simboli | NR3C1; GCCR; GCR; GR; GRL | ||||||||||
| Vanjski ID | OMIM: 138040 MGI: 95824 HomoloGene: 30960 IUPHAR: GeneCards: NR3C1 Gene | ||||||||||
| |||||||||||
| Pregled RNK izražavanja | |||||||||||
 | |||||||||||
 | |||||||||||
 | |||||||||||
| podaci | |||||||||||
| Ortolozi | |||||||||||
| Vrsta | Čovek | Miš | |||||||||
| Entrez | 2908 | 14815 | |||||||||
| Ensembl | ENSG00000113580 | ENSMUSG00000024431 | |||||||||
| UniProt | P04150 | E9PUR6 | |||||||||
| RefSeq (mRNA) | NM_000176 | NM_008173 | |||||||||
| RefSeq (protein) | NP_000167 | NP_032199 | |||||||||
| Lokacija (UCSC) |
Chr 5: 142.66 - 142.82 Mb |
Chr 18: 39.41 - 39.49 Mb | |||||||||
| PubMed pretraga | [1] | [2] | |||||||||
Glukokortikoidni receptor (GR, GCR, NR3C1, nuklearni receptor potfamilije 3, grupa C, član 1) receptor je za koji se vezuju kortizol i drugi glukokortikoidi.
GR je izražen u skoro svim ćelijma a telu i reguliše gene koji kontrolišu razviće, metabolizam, i imunski respons. Gen ovog receptora se izražava u nekoliko formi, te stoga GR ima mnoštvo različitih (pleiotropskih) dejstava u različitim delovima tela.
Kad se za GR vežu glukokortikoidi, njegov primarni mehanizam dejstva je regulacija trakripcije gena.[1][2] Nevezani receptor se nalazi u ćelijskom citosolu. Nakon vezivanja glukokortikoida za receptor, receptor-glukortikoidni kompleks može da povisi izražavanje antiinflamatornih proteina u jedru ili da suzbije izražavanje proinflamatornih proteina u citozolu (putem sprečavanja translokacije drugih transkripcionih faktora iz citozola u jedro).
Kod ljudi, GR protein je kodiran NR3C1 genom koji je lociran na hromozomu 5 (5q31).[3][4]