Howlers corrected
- Solubility equilibrium many solids will become more soluble with increasing temperature, regardless of Le Chatelier's Principle
- pH the units of pH are log(L/mol), although this is seldom indicated explicitly
- Phosphoric acid There is an increased risk of fire or causing an explosion from adding strong oxidizers to phosphoric acid.
- Frigorific mixture When ammonium chloride salt is placed on ice...heat is absorbed and the temperature rises.
- Lanthanide The colors of lanthanide complexes originate almost entirely from charge transfer interactions between the metal and the ligand.
- Glucose .. has a dissociation exponent (pK) of 12.16 at 25 °C (77 °F) in methanol and water The same value in two solvents?
- Copper(II) sulfate Anhydrous copper(II) sulfate is a white solid.
- Hydrochloric acid is the salt of the protonated water and chloride
- Amoxicillin was [] developed at Beecham, England. In this context Beecham is the name of a company, not a place!
- Ibuprofen It is an optically active compound. Each isomer is optically active. The commercial product with this name is a racemic mixture of the two isomers.
- Azurite is... a carbonate with the chemical formula Cu3(CO3)2(OH)2 Astonishing! the formula clearly shows that it is a basic carbonate and the text states (below) that copper carbonate is not known.
- Isomerizations with low energy difference... are endothermic. The sign of the free energy difference depends on how the reaction is defined, A⇌B or B⇌A.
- Ammonium nitrate has the chemical formula NH
4NO
3, simplified to N2H4O3! - Chemistry of ascorbic acid Ascorbic acid is a vinylogous carboxylic acid It does not contain a carboxyl group
- Properties of water#Chemical properties water dissociates disproportionately into a hydronium ion and hydroxide ion. A brilliant Malapropism.
- Chemistry When hydrogen bromide is dissolved in water it forms hydrobromic acid. It is not a dissolution reaction. There is a chemical reaction: HBr(g) + H2O(l) → H3O+(aq) + Br-.
- Americium Am3+ is colorless at low and reddish at higher concentrations. Confusion between intrinsic colour and colour intensity.
- Trimethylglycine ...exists as the zwitterion at neutral pH. The zwitterion is present in solution over the range pH > (pKa-2). The same error is present in Betaine
- Spermidine Polyamines, such as spermidine, are polycationic aliphatic amines neutral molecule
- Buffering agent the function of a buffering agent is to prevent a rapid change in pH... It's nothing to do with speed.
- Lactose yogurt ... have lactose contents similar to that of milk.
- 6-APA In 1958, Beecham scientists in the UK discovered 6-APA. What they actually did was to find a way to chemically isolate 6-APA from penicillin so that new antibiotics could be synthesized by attaching other side-chains to the nucleus and new antibiotics like ampicillin could be created from it. N.b. 6-APA is a beta lactam and this name is used for the class of β-lactam antibiotics derived from it, including penicillin.
- Sodium pyrosilicate is ... a salt of the unstable pyrosilicic acid. The acid is stable in non-aqueous solution. In aqueous solution it will be in equilibrium with SiO2 which is insoluble in water.
- Lithium cobalt oxide The space group is therefore ... Symmetry is a property of the space group.
- Solar cell#theory Photons … are absorbed by semiconducting materials, such as silicon. Pure silicon is not a semi-conductor.
- Lead(II,IV) oxide Lead tetroxide is most often used as... Pb3O4 is not a tetroxide!
- Hydrofluoric acid Foremost are cryolite and aluminium trifluoride. A molten mixture of these solids… A liquid mixture of these compounds
- Hydrofluoric acid the ionization [of HF] can be described as a pair of successive equilibria. The equilibria are simultaneous, not successive.
- Antimony [The element] reacts with oxygen to produce antimony trioxide, Sb2O3. The formula clearly shows that it is a sesquioxide.
- Quinhydrone electrode Quinhydrone [dissolves] to form a mixture of two substances at equal concentration. The electrode function depends on the concentration ratio varying with pH!
- Lithium-ion battery lithium ... ignites when exposed to water. Lithium metal reacts with water to produce "nascent hydrogen" which is flammable
- tophus is a deposit of uric acid crystals, in the form of monosodium urate crystals
- Chrome orange consists of lead(II) chromate and lead(II) oxide. Pb2CrO5 is a mixed oxide
- Lead(II) nitrate Structure
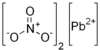 removed. It shows only one canonical form of a resonance hybrid and gave a misleading impression that the nitrate ion is not trigonally symmetric.
removed. It shows only one canonical form of a resonance hybrid and gave a misleading impression that the nitrate ion is not trigonally symmetric. - Ammonium dihydrogen phosphate Ammonium dihydrogen phosphate … with formula NH
6PO
4. The empirical formula is meaningless in this context. - Nitric acid#Structure and bonding The [nitric acid] molecule is planar. Not so; the OH group is free to rotate except when the molecule is in the solid state. The nitrate ion is planar.
- Beryllium Currently, most beryllium is produced by reducing beryllium fluoride with purified magnesium How can an element be purified?
- Water oxidation catalysis Some metal hydroxides... can oxidize to give metal oxo complexes. Can be oxidised!
- Indium tin oxide ITO is a heavily doped n-type semiconductor. The whole section is garbled. With a ratio In:Sn of ca. 4:1 this substance is best described as a solid solution of tin oxide in indium oxide. However the term "doped" seems to be in common use, so it will be retained.
- Lithium soap The most useful of the non-detergent soaps are... Wonderful contradiction of terms
- Saltwater soap Potassium is a alkali metal similar to sodium but more active
- Phenol reacts completely with aqueous NaOH to lose H+ Missing the word excess, otherwise the reaction is incomplete.
- Magnesium acetate [Synthesis] Reacting metallic magnesium with acetic acid dissolved in dry benzene causes magnesium acetate to form along with the release of a gas, presumably hydrogen - is followed by the correct equation!
- Calcium acetate pKa=6.3 - 9.6. The average of a dozen published values is 0.7±0.4. The higher values, though reported in the literature are completely fallacious.
- Metal aquo complex [V(H2O)6]5+ is unknown, but its conjugate base, [VO(H2O)5]2+ is ... Wrong electrical charge! Hydrolysis does not involve a change in oxidation state.
- 8-Hydroxyquinoline is a monoprotic bidentate chelating agent. Not so: the chelating agent is the hydroxyquinolate anion.
- Germanium Featured article! Elemental germanium oxidizes slowly to GeO2 at 250 °C. In air?. This reaction cannot occur at a precise temperature.
- Aluminium smelting Alumina is dissolved in molten cryolite, typically at 960 °C, according to the following simplified reactions:Al2O3 + 3⁄2C ⇌ 2Al + 3⁄2CO2. Completely garbled! The reduction is an electrolytic process, during which the carbon electrode may be oxidised.
- Blast furnace Carbon monoxide also reduces silica ??
- Acid dissociation constant the activity of solvent water using pure water as a standard state is approximated as one Confused activity with activity coefficient
- Properties of water Water... has its own intrinsic very light blue hue. Ice also appears colorless! An amazing contradiction!
- Fusion power Tritium is a natural isotope of hydrogen. It has too short a half-life (ca. 12 y) to exist in nature.
- Francium is the second-least electronegative element. It is electropositive
- Metal ions in aqueous solution the molybdenum(IV) species formulated as [(H4O)-Mo-Mo-(OH4]4+ Wrong change of oxidation state from II to IV.
- Ionization energy is qualitatively defined as... I.E. is a quantitative measure.
- Solvatochromism Due to the Franck–Condon principle (atoms do not change position during light absorption — except chromophores
- Dodecaborate is an ionic molecule... Is it an ion or is it a molecule? (The term "molecular ion" is used in mass spectrometry for a molecule that has lost 1 electron)
- Carborane The concentration of H+ in a solution of the compound H(CHB11Cl11) is a million times higher than in a solution of sulfuric acid. Confusing concentration with pK value
- Phosphoric acid … is a mineral and weak acid. Phosphoric acid is not a mineral
- Phosphoric acid There is an increased risk of fire or causing an explosion from adding strong oxidizers to phosphoric acid. P(V) cannot be oxidised.
- Acid strength(version 8 July 2018) acids become stronger as they are diluted. For a monobasic acid the proportion of free to total acid concentrations is dependent only on pH .
- Ortho acids In general, when an element can form several oxoacids ... with the ortho acid being the most hydrated of the three. A compound formed by an hydration reaction such as P2O5 + 3H2O → 2H3PO4 is not a hydrate, as it contains no water molecules.
- The history of aspirin stretches back to antiquity. Salycilic acid, yes. Aspirin (Acetylsalycilic acid), no.
- Metasilicic acid ... H
2SiO
3... is one of the most common silicic acids. No compound with this chemical, formula is known. - Metaphosphoric acid is a singly anhydrous version of orthophosphoric acid
- Enthalpy of neutralization The standard enthalpy change of neutralization for a strong acid and base is -57.62 kJ/mol.No temperature specified
- nickel hydroxide In the chembox(solubility) it is given as "soluble in dilute acid". Nickel hydroxide reacts with acid to produce a solution containing Ni2+ ions, not the hydroxide in solution.
- magnesium hydroxide Synthetic magnesium hydrides is often known as milk of magnesia a brilliant typo.
- Ionization energy Successive molar ionization energies in kJ/mol (96.485 kJ/mol = 1 eV/particle)
- Sodium orthovanadate is the inorganic compound with the formula Na3VO4(H2O)2. It is one of several vanadium(V) oxides !
- Elemental magnesium ... unlike the other alkali metals Unbelievable!
- Osmosis The osmotic pressure is defined to be the minimum pressure required to maintain an equilibrium minimum!
- 18-Crown-6 ... wraps around metal cations, providing an octahedral cavity. It is 6-coordinate, not octahedral.
- Niacin pka=4.85. This pK refers to the protonation of the N atom. The carboylic acid part of the molecule has a pKa of ca. 2. This is a howler because only a single value was cited.
- Helium ...it exists only as a gas except in extremely cold conditions. Meaningless! Phrase removed.
 Saponification The alkoxide is more basic than the conjugate base of the carboxylic acid, and hence proton transfer is rapid. :An example of confusion between an equilibrium property, basicity, and a kinetic property, reaction rate.
Saponification The alkoxide is more basic than the conjugate base of the carboxylic acid, and hence proton transfer is rapid. :An example of confusion between an equilibrium property, basicity, and a kinetic property, reaction rate.- Chlorite dismutase ..the two substrates of this enzyme are chloride ion and oxygen, whereas its product is chlorite. It's the other way round - oxygen is a product. This error is present in the enzyme catalogue (EC 1.13.11.49), from which, presumably, it was copied.
- Nitrogenase Dinitrogen is quite inert because of the strength of its N≡N triple bond. What is astonishing about this error is that it is followed by a correct statement relating inertness to high activation energy.
- Glucose Like all hexoses, glucose has the molecular formula C6H12O6. It has fifteen stereoisomers. Glucose has 15 isomers? What nonsense!
- Sulfur trioxide#Structure and bonding (Version 14 Aug. 2014) As sulfur trioxide is centrosymmetric , it has no electrical dipole.The molecule cannot have a centre of symmetry, as it has 3-fold symmetry
- Metal ions in aqueous solution version 24 Sept.2013. Raman spectroscopy is particularly useful because the excitation wavelength for the Raman spectra could be choosen in the region of water's transparancy. Nonsense! It's nothing to do with excitation wavenength. Reverted
- Savitzky-Golay smoothing filter, version 09:57, 30 May 2013. Omitted value of the data interval in the expressions for derivatives.
- Radioactive tracer Phosphorus-32 is frequently used to label amino acids. Revised
- Radioactive tracer Tritium and carbon-14-labeled water and glucose are commonly used.. Revised
- Fractional crystallization (chemistry) [In] fractional crystallization [of] two or more substances they will crystallize out of solution (precipitate) at different rates. It is not a matter of rates, but of different solubility products which determine the equilubrium state.
- polyphosphate: .. are anionic phosphate polymers linked between hydroxyl groups and hydrogen atoms. Article extensively revised
- EDTA is an amino acid. Replaced by polyamino carboxylate.
- Lutetium is a d-block element In transition metal, version 2 Aug 2009
- Caption to image "basic zinc acetate.png" labelled zinc acetate. Featured article zinc!!!
- Niobium is in many ways similar to its predecessors in group 4. Featured article niobium!!! Changed to group 5.
- metaphosphate A metaphosphate is a salt or an ester of metaphosphoric acid, HPO3
- reduction potential The half cell standard potential is given by where Δ G is the Gibbs free energy change. Changed to standard free energy change.
- Crystal field excitation dd-transitions on a copper atom that is surrounded by a hexagon of oxygen atoms. Changed in Revision as of 19:38, 17 May 2010 to an octahedra. Finally changed to an octahedron.
- Recrystallization (chemistry) Since "Compound A" is in excess this will usually result in these crystals forming first see fractional crystallization, above
- Molecular vibration (version 19 December 2010) The three atomic molecule have three modes of vibration irrespective of whether these are linear or nonlinear. Confuses normal modes of vibration with vibration frequencies.
- Hydroxide (version 5 December 2010) When hydroxide ion is mixed with hydrogen gas, water forms. Article re-written.
- uranyl: (Version 23 Jan 2011) Examples of uranyl compounds include uranium trioxide Whole article revised.
- Technetium-99m(Earlier version):The molybdenum-99 is in the form of sodium molybdate in ion resins. When the Mo-99 decays to Tc-99m this does not form a similar compound Corrected by another editor.
- Borate the fictitious ions [ .. H2BO−
3, HBO2−
3 and BO3−
3] are easier to use for pKa values and other calculations. Article revised. - Uranate Uranate is the chemical term for oxide anions of the element uranium. Examples of uranates include UO22-, UO32−, and UO42− Article re-written.
- pH "The meaning of the "p" in "pH" is unknown". No it isn't. It means decimal cologarithm
- pH "where aH+ is the activity of hydrogen ions in units of mol/L (molar concentration)" Logarithms can only be taken of numbers, so the idea of taking a logaritm of a concentration is nonsense. In fact activity is dimensionless.
- Electromagnetic absorption by water Rotational transitions don't affect significantly the absorption spectrum of water. Complete omission of vibration-rotation effects. Article re-wrtten
- Buffer solution (Revision as of 08:43, 6 May 2013) most of the added hydroxide ion is consumed in the reaction OH- + HA → H2O + A- ... which results in a decrease in pH. Corrected by user:Edgar181
- Borax (as of 1 Oct 2013)... is a salt of boric acid. changed to with an empirical formula Na2B4O7nH2O•
- Sodium hydroxide (As of 1 Oct 2013) Sodium hydroxide slowly reacts with glass to form sodium silicate. Glass dissolves, forming soluble silicates.
- Ascorbic acid (As of 1 Oct 2013) [..] behaves as a vinylogous carboxylic acid. Re-written in terms of electron delocalization stabilizing the ascorbate anion.
- Fluorinated ethylene propylene ... initiated with persulfate, which homolyzes to generate sulfate radials. A brilliant typo!
- Denticity: EDTA is a unique ligand which forms a chelate by binding to a central metal at six different points. Removed by another editor.