Structure
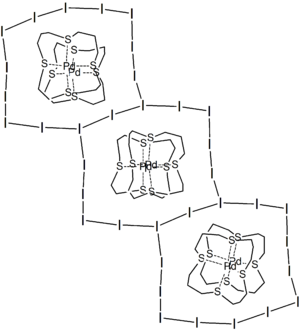

Polyiodides adopt diverse structures. Most can be considered as associations of I2, I−, and I−
3 units. Discrete polyiodides are usually linear. The more complex two- or three-dimensional network structures of chains and cages are formed as the ions interact with each other, with their shapes depending on their associated cations quite strongly, a phenomenon named dimensional caging.[4][5] The table below lists the polyiodide salts which have been structurally characterized, along with their counter-cation.[6]
| Anion | Counter-cation | Structural description |
|---|---|---|
| [I2]− | Na(C3H6O)+ 3 |
linear[7][8] |
| [I3]− | Cs+, (C4H9)4N+ | linear |
| [I4]2− | [Cu(NH3)4]2+ | symmetric linear array of iodine atoms[9] |
| [I5]− | [EtMe3N]+ | V-shaped with polymeric layers |
| [EtMePh2N]+ | V-shaped with isolated [I5]− ions | |
| [I6]2− | [NH3(CH2)8NH3]2+ | almost linear [[10]] |
| [I7]− | [Ag(18aneS6)]+ | an anionic network derived from a primitive rhombohedral lattice of iodide ions bridged by I2 molecules |
| [I8]2− | [Ni(phen)3]2+ | regular anionic shapes, can be described as [I− 3·I2·I− 8] or [I− 3·I− 5] |
| [I9]− | [Me2iPrPhN]+ | 14-membered ring tied by two I2 bridges to give 10-membered rings |
| [Me4N]+ | non-octahedral, but a twisted "h"-like arrangement of I− 3 and I2 units | |
| [I10]2− | [Cd(12-crown-4)2]2+; Theophyllinium | twisted ring configuration with two I− 3 units linked by two I2 molecules[11] |
| [I11]3− | [(16aneS4)PdIPd(16aneS4)]3+ | 14-membered ring (9.66 × 12.64 Å) around the complex cation, with the rings interlink further to give an infinite 2D sheet |
| [I12]2− | [Ag2(15aneS5)2]2+ | extended 3D spiral superstructure supported by Ag–I bonds and weak I···S interactions |
| [Cu(Dafone)3]2+ | planar configuration | |
| [I13]3− | [Me2Ph2N]+ | consists of zigzag chains of I− and I2 |
| [I14]4− | 4,4′-bipyridinium | double hook (I− 3·I2·I−·I2·I−·I2·I− 3)[12] |
| [I16]2− | [Me2Ph2N]+ | centrosymmetric arrangement of [I− 7·I2·I− 7] |
| [iPrMe2PhN]+ | the anion forms 14-membered rings catenated by I2 molecules, which further link into layers with 10- and 14-membered rings | |
| [I22]4− | [MePh3P]+ | two L-shaped [I5]− units linked by an I2 molecule and completed by two end-on [I5]− groups |
| [I26]3− | [Me3S]+ | consists of [I5]− and [I7]− ions with intercalated I2 molecules |
| [I26]4− | Cp*2Fe+ | an anionic network derived from a primitive cubic lattice built from I− ions, with I2 bridges on all edges and systematically removing 1⁄12 of the I2 molecules |
| [I29]3− | Cp2Fe+ | an anionic 3D network with a cage-like structure of [{(I− 5)1⁄2·I2}·{(I2− 12)1⁄2·I2}·I2], with [Cp2Fe]+ ions interacting with the anion in the cavities[13] |
| [I∞]δ− | Pyrroloperylene+• | Infinite polyiodide homopolymer.[14] |
