| |
| Names | |
|---|---|
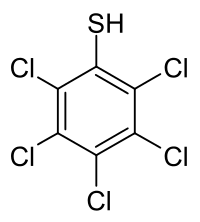
| Preferred IUPAC name
Pentachlorobenzenethiol | |
| Other names
Pentachlorobenzenethiol, pentachlorothiophenol, PCBT
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.004.644 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6HCl5S | |
| Molar mass | 282.38 g·mol−1 |
| Appearance | Gray solid |
| Density | 1.7±0.1 g/cm3 |
| Melting point | 231.5 °C (448.7 °F; 504.6 K) |
| Insoluble | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| Flash point | 144.6 °C (292.3 °F; 417.8 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentachlorobenzenethiol is a chemical compound from the group of thiols and organochlorine compounds.[1] The chemical formula is C
6HCl
5S.[2]