| |
| Names | |
|---|---|
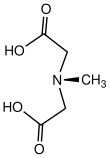
| Other names
N-(Carboxymethyl)-N-methyl-glycine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.022.326 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H9NO4 | |
| Molar mass | 147.130 g·mol−1 |
| Appearance | white solid |
| Melting point | 223–225 °C (433–437 °F; 496–498 K) |
| Hazards | |
| GHS labelling:[1] | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
N-Methyliminodiacetic acid is an organic compound with the formula CH3N(CH2CO2H)2. It is a white solid, which as its conjugate base CH3N(CH2CO−2)2 is used as a chelating agent for iron.[2] It is a component of organoboron reagents as well.[3]