| |
| Names | |
|---|---|
| IUPAC name
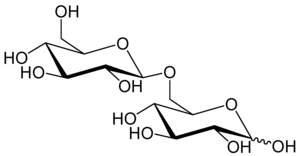
6-O-β-D-glucopyranosyl-D-glucose
| |
| Other names
amygdalose
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H22O11 | |
| Molar mass | 342.30 g/mol |
| Density | 1.768 g/mL |
| Melting point | 190 to 195 °C (374 to 383 °F; 463 to 468 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Gentiobiose is a disaccharide composed of two units of D-glucose joined with a β(1->6) linkage. It is a white crystalline solid that is soluble in water or hot methanol. Gentiobiose is incorporated into the chemical structure of crocin, the chemical compound that gives saffron its color. It is a product of the caramelization of glucose.[2] During a starch hydrolysis process for glucose syrup, gentiobiose, which has bitterness, is formed as an undesirable product through the acid-catalyzed condensation reaction of two D-glucose molecules.[3] One β-D-glucose unit elongation of the bitter disaccharide reduces its bitterness by a fifth, as determined by human volunteers using the trimer, gentiotriose.[4] Gentiobiose is also produced via enzymatic hydrolysis of glucans, including pustulan[5] and β-1,3-1,6-glucan.[6]