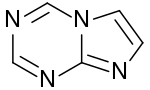
| |
| Names | |
|---|---|
| Preferred IUPAC name
Imidazo[1,2-a][1,3,5]triazine | |
| Other names
5-Aza-7-deazapurine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C5H4N4 | |
| Molar mass | 120.115 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
5-Aza-7-deazapurine or imidazo[1,2-a][1,3,5]triazine[1] is a heterocyclic aromatic organic compound that consists of a s-triazine ring fused to an imidazole ring. It is an isostere and isomer of purine. However, in 5-aza-7-deazapurine, N-9 of five-membered ring does not bond with hydrogen. So 5-aza-7-deazapurine derivatives must have an exocyclic substituent with a double bond to bind a sugar residue.[2] 5-Aza-7-deazapurine nucleosides may have an oxo, thioxo, or a imine group.
Notable derivatives of this molecule include 5-aza-7-deazaguanine,[2] which is a nucleobase of hachimoji DNA.