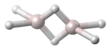| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Dialumane(6)
| |||
| Other names
Dialumane, dialane
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| |||
| |||
| Properties | |||
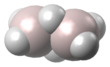
| Al2H6 | |||
| Molar mass | 60.011 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Dialane is an unstable compound of aluminium and hydrogen with formula Al2H6.[1][2][3] Dialane is unstable in that it reacts with itself to form a polymer, aluminium hydride. Isolated molecules can be stabilised and studied in solid hydrogen.[2]